CAS Number 554-13-2, Lithium Carbonate or Dilithium Carbonate USP Grade Manufacturers Exporters







CAS Number 554-13-2, Lithium Carbonate or Dilithium Carbonate Manufacturer Exporter
For Properties Specifications of Lithium Carbonate or Dilithium Carbonate Click Properties, Specifications of Lithium Carbonate or Dilithium Carbonate Manufacturer.
For Uses of Lithium Carbonate or Dilithium Carbonate Click Uses of Lithium Carbonate or Dilithium Carbonate Manufacturer.
For For SDS MSDS Sheet of Lithium Carbonate or Dilithium Carbonate Click SDS Safety Data Sheet MSDS Sheet of Lithium Carbonate or Dilithium Carbonate Manufacturer.
The Properties and Specifications of Lithium Carbonate or Dilithium Carbonate:
Appearance: White powder.
Melting point: 723C literature.
Boiling point: 1310C (decomposes) literature.
Relative density:
2.1
Lithium Carbonate USP Grade Specifications:
Li2CO3 - 73.89
Carbonic acid, dilithium salt
Dilithium carbonate CAS 554-13-2
DEFINITION
Lithium Carbonate contains NLT 99.0% of lithium carbonate (Li2CO3), calculated on the dried basis.
IDENTIFICATION
A. It effervesces upon the addition of an acid, yielding a colorless gas that, when passed into calcium hydroxide TS, immediately forms a white precipitate.
B. When moistened with hydrochloric acid, it imparts an intense crimson color to a nonluminous flame.
Aluminum and Iron
Sample solution: Dissolve 500 mg of Lithium Carbonate in 10 mL of water by the dropwise addition, with agitation, of hydrochloric acid.
Analysis: Boil the Sample solution, then cool it. To 5 mL of the solution add 6 N ammonium hydroxide until the reaction is alkaline.
Acceptance criteria: No turbidity or precipitate is observed.
Calcium: To pass the test. (0.15%).
Sodium: To pass the test. (0.1%).
Chloride and Sulfate, Sulfate: To pass the test. (0.1%).
Chloride and Sulfate, Chloride: To pass the test. (0.07%).
Loss on Drying: Dry at 200C for 4 h. NMT 1.0%
Packaging and Storage: Preserve in well-closed containers.
Specifications of Lithium Carbonate BP Li2CO3 Grade
Li2CO3 --- 73.9 --- CAS 554-13-2
DEFINITION
Content: 98.5 per cent to 100.5 per cent.
CHARACTERS
Appearance: White or almost white powder.
Solubility: Slightly soluble in water, practically insoluble in ethanol (96 per cent).
IDENTIFICATION
A. When moistened with hydrochloric acid, it gives a red colour to a non-luminous flame.
B. Dissolve 0.2 g in 1 ml of hydrochloric acid. Evaporate to dryness on a water-bath. The residue dissolves in 3 ml of ethanol (96 per cent).
C. It gives the reaction of carbonates.
TESTS
Solution S: Suspend 10.0 g in 30 ml of distilled water and dissolve by the addition of 22 ml of nitric acid. Add dilute sodium hydroxide solution until the solution is neutral and dilute to 100 ml with distilled water.
Appearance of solution: Solution S is clear and colourless.
Chlorides: Maximum 200 ppm.
Sulphates: Maximum 200 ppm.
Arsenic: Maximum 2 ppm, determined on 0.5 g.
Calcium: Maximum 200 ppm.
Iron: Maximum 20 ppm.
Magnesium: Maximum 150 ppm.
Potassium: Maximum 300 ppm.
Sodium: Maximum 300 ppm.
The Uses of Lithium Carbonate or Dilithium Carbonate:
Lithium Carbonate is used to treat the symptoms of Bipolar Disorder. It works to stabilize the mood and reduce extremes in behavior by restoring the balance of certain natural substances in the brain. It is widely used in the processing of metal oxides. Lithium carbonate is used in ceramic glaze. Its main use is as a precursor for compounds used in lithium-ion batteries. It is an important industrial chemical. It forms low-melting fluxes with silica and other materials. Cement sets more rapidly when prepared with lithium carbonate, and is useful for tile adhesives. It is used as an active material of carbon dioxide sensors.The MSDS-SDS Hazard Statement of Lithium Carbonate or Dilithium Carbonate:
Lithium Carbonate or Dilithium Carbonate GHS SDS, Safety Data Sheet
MSDS
1. Product Identification
Product Name & Other Names : Lithium Carbonate or Carbolithium or Dilithium Carbonate or Carbonic acid lithium salt
CAS No.: 554-13-2
EINICS EC-No.: 209-062-5
Relevant uses and uses advised against (if any) : Industrial Manufacturing.
Suppliers: As per letterhead.
2. Hazards Identification
GHS, Globally Harmonized System Classification in accordance with 29 CFR 1910
Classification according to Regulation (EC) No 1272/2008
Acute toxicity, oral Category 4, H302
Serious eye damage/eye irritation Category 2A, H319
Labeling according to GHS & Regulation (EC) No 1272/2008
GHS Label Elements Irritant |
Signal Words: Warning
Hazard statements:
H302: Harmful if swallowed.
H319: Causes serious eye irritation.
Precautionary statements:
P262: Do not get in eyes, on skin, or on clothing.
P264: Wash skin thoroughly after handling.
P280: Wear protective gloves/protective clothing/eye protection/face protection.
P270: Do not eat, drink or smoke when using this product.
P330: Rinse mouth.
P301+312: IF SWALLOWED: Call a POISON CENTER or doctor/physician if you feel unwell.
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing.
P337+P313: If eye irritation persists: Get medical advice/ attention.
3. Composition/Information on Ingredients
Product Name & Other Names: Lithium Carbonate or Carbolithium or Dilithium Carbonate or Carbonic acid lithium salt
CAS No.: 554-13-2
EINICS EC-No.: 209-062-5
4. First Aid Measures
Always get medical attention after the first aid is over.
Inhalation: If inhaled, remove to fresh air. If not breathing, give artificial respiration. If breathing is difficult, give oxygen and get medical attention immediately.
Ingestion: Never give anything by mouth to an unconscious person. Get medical attention.
Skin Contact: Immediately flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Get medical attention. Wash clothing and shoes before reuse.
Eye Contact: Immediately flush eyes with plenty of water for at least 15 minutes, lifting lower and upper eyelids occasionally. Get medical attention immediately.
5. Fire Fighting Measures
Fire: Not considered as a fire hazard.
Fire Extinguishing Media: Water spray, dry chemical, alcohol foam, or carbon dioxide.
Extinguishing Media Not recommended: Avoid using solid water jet as it may scatter the fire.
Special Information: In the event of a fire, wear full protective clothing and NIOSH-approved self-contained breathing apparatus with full face piece operated in the pressure demand or other positive pressure mode. At elevated temperatures under fire conditions, it may produce toxic or irritating fumes. Fire-extinguishing work is done from the windward and the suitable fire-extinguishing method according to the surrounding situation is used. Uninvolved persons should evacuate to a safe place.
6. Accidental Release Measures
Personal precautions, protective equipment, and emergency procedures: Ventilate area of leak or spill. Avoid breathing dust/fumes/gas/mist/vapors/spray. Use individual protective equipment (waterproof boots, suitable protective clothing, safety glasses, etc.). Prevent any contact with hot surfaces. Do not approach facing the wind.
Environmental precautions: Do not let the product enter drains, soil, or water sources.
Methods and materials used for containment cleanup procedures and Storage: Contain spilled material. Cover with an inert, non-combustible absorbent material, (e.g., sand, earth, diatomaceous earth, vermiculite). Vacuum or sweep-up and remove to an approved disposal container. Finish cleaning by spreading water on the contaminated surface and allow to evacuate as per law.
7. Handling and Storage
Precautions for safe handling: Apply according to good manufacturing and industrial hygiene practices. Ensure proper ventilation.In case of insufficient ventilation, wear suitable respiratory equipment. Wash thoroughly after handling. Do not drink, eat, or smoke while handling. Avoid contact with skin, eyes, and clothing. Minimize dust generation. Avoid breathing dust/fumes/gas/mist/vapors/spray. Use individual protective equipment (waterproof boots, suitable protective clothing, safety glasses, etc.). Prevent any contact with hot surfaces.
Conditions for safe storage, including any incompatibilities: Store in cool, dry, and ventilated area away from heat sources and protected from sunlight in tightly closed original container. Keep air contact to a minimum. Store protected from heat, sparks and ignition sources and incompatible materials. Do not store with incompatible materials like strong oxidizing agents and acids.
8. Exposure Controls/Personal Protection
Airborne Exposure Limits: OSHA-PELs TWA: 15 (Total dust), 5 (Respirable fraction) as Particulates not otherwise classified
Ventilation System: A system of local and/or general exhaust is recommended to keep employee exposures as low as possible.
Personal Respirators (NIOSH Approved): For conditions of use where exposure to dust or mist is apparent and engineering controls are not feasible, a particulate respirator (NIOSH type N95 or better filters) may be worn.
Skin Protection: Wear impervious protective clothing, including boots, gloves, lab coat, apron, or coveralls, as appropriate, to prevent skin contact.
Eye Protection: Use chemical safety goggles and/or full-face shield where dusting or splashing of solutions is possible. Maintain eye wash fountain and quick-drench facilities in work area.
Other Control Measures: Maintain good housekeeping in work area. Handle in accordance with good industrial hygiene and safety practice. Wash hands after handling.
9. Physical and Chemical Properties
Appearance: White to off-white crystals or powder.
Odor: Odorless.
Odor threshold: No data found.
pH: Approximately 9-11.
Relative density: around 1.6
Melting Point: 618C - literature.
Initial boiling point and boiling range: No data found.
Flash point: No data found.
Auto-ignition temperature: No data found.
Decomposition temperature: No data found.
Upper/lower flammability or explosive limits: No data found.
Vapor pressure: No data found.
Vapor density: No data found.
Evaporation rate: No data found.
Flammability (solid, gas): No data found.
Partition coefficient: n-octanol/water: No data found.
Solubility: Slightly soluble in water.
Viscosity: No data found.
Molecular Weight: 73.89
Chemical Formula: Li2CO3
10. Stability and Reactivity
Stability: Stable under ordinary conditions of use and storage.
Hazardous Decomposition Products: Carbon dioxide and carbon monoxide along with lithium oxide may form when heated to decomposition.
Hazardous Polymerization: Will not occur.
Incompatibilities: Excessive heat, Strong oxidizers, and acids.
Conditions to Avoid: Heat, dusting and incompatibles.
11. Toxicological Information
LD50 Oral - Rat - 525 mg/kg(Lithium carbonate)
LC50 Inhalation - Rat - 4 h - > 2.17 mg/l(Lithium carbonate)
Carcinogenicity: No component of this product present at levels greater than or equal to 0.1% is identified as possible or confirmed human carcinogen by IARC, ACGIH, OSHA and NTP.
Mutagenic Effects: No data found.
Teratogenic Effects: No data found.
Developmental Toxicity: No data found.
Reproductive Effects: No data found.
12. Ecological Information
Toxicity to fish: LC50 - Oncorhynchus mykiss (rainbow trout) - 30.3 mg/l - 96 h
Toxicity to daphnia and other aquatic invertebrates: EC50 - Daphnia magna (Water flea) - 33.2 mg/l - 48 h
Mobility in soil: No data found
PBT & vPvB: This substance/mixture contains no components considered to be either persistent, bioaccumulative and toxic (PBT), or very persistent and very bioaccumulative (vPvB) at levels of 0.1% or higher.
13. Disposal Considerations
Whatever cannot be saved for recovery or recycling should be managed in an appropriate and approved waste disposal facility.
14. Transport Information
DOT USA, TDG Canada & ADR/RID Europe: Not controlled for transport.
IATA/ICAO: Not controlled for transport.
IMDG/IMO: Not controlled for transport.
15. Regulatory Information
USA:
California Proposition 65: Not listed.
SARA 311/312 Hazards: See section 2.
16. Other Information
European Labeling in Accordance with EC Directives:
H302 = Harmful if swallowed.
H319 = Causes serious eye irritation.
Classification according to EU Directives 67/548/EEC or 1999/45/EC:
Hazard Symbol:
Xi = Irritant
Risk Phrases:
R22 Harmful if swallowed.
R36= Irritating to eyes.
***********************************
Our company provides this Lithium Carbonate MSDS sheet in good faith but makes no representation as to its comprehensiveness or accuracy. This SDS sheet is intended only as a guide to the appropriate precautionary handling of the material by a professionally trained person using this product. The above information has been compiled from various sources and has the possibility of discrepancy and being outdated information. Individuals receiving the information must exercise their independent judgment and do further search in determining its appropriateness for a particular purpose. In no case shall our company be liable to loss or damages by the product user.
***********************************
Anmol Chemicals Industries Ltd. is an off-shoot of Anmol Chemicals and Anmol Chemicals & Pharmaceuticals, Taloja. It is located in MIDC Taloja and it is manufacturing pharmaceutical grades of API, Excepients, Food grade and Reagent grade chemicals. Anmol Chemicals Industries Pvt. Ltd. is a several decades old group of companies, engaged in manufacturing, supplying, distributing, wholesale supplies for actual users, retail or small pack supplies for research and development chemicals, fine and speciality chemicals, pharmaceutical excipients, mineral fortifiers in chemically pure, Analytical reagent grade, IP BP USP Ph Eur EP JP and other pharmaceutical grade monograph including FCC Food grade chemicals and Nutraceuticals, Mineral Fortifiers at best prices.
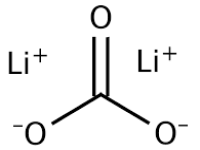
Lithium Carbonate or Dilithium Carbonate Structure
CAS Number 554-13-2, Lithium Carbonate or Dilithium Carbonate Manufacturer Exporter
ANMOL CHEMICALS INDUSTRIES Pvt. Ltd.
India, USA, Europe, UAE
TELEPHONE: 91-22-65290110
Navi Mumbai, INDIA
e-mail: info(At the rate i.e. @)anmol.org
Copyright. 24-may-26
We manufacture:
Glacial Acetic Acid Manufacturer
Dihydroxyaluminum Sodium Carbonate

