CAS Number 9067-32-7, Sodium Hyaluronate USP NF BP Ph Eur Purified Sodium Hyaluronate JP Grade Manufacturers Exporters







CAS Number 9067-32-7, Sodium Hyaluronate USP NF BP Ph Eur, Purified Sodium Hyaluronate JP Manufacturer Exporter
For Properties Specifications of Sodium Hyaluronate Click Properties, Specifications of Sodium Hyaluronate Manufacturer.
For Uses of Sodium Hyaluronate Click Uses of Sodium Hyaluronate Manufacturer.
For For SDS MSDS Sheet of Sodium Hyaluronate Click SDS Safety Data Sheet MSDS Sheet of Sodium Hyaluronate Manufacturer.
The Properties and Specifications of Sodium Hyaluronate:
Sodium Hyaluronate BP Ph Eur Grade Specifications
Ph Eur monograph
(C14H20NNaO11)n --- CAS 9067-32-7
Action and use: High viscosity mucopolysaccharide.
DEFINITION
Sodium salt of hyaluronic acid, a glycosaminoglycan consisting of D-glucuronic acid and Nacetyl- D-glucosamine disaccharide units.
Content: 95.0 per cent to 105.0 per cent (dried substance).
Intrinsic viscosity: 90 per cent to 120 per cent of the value stated on the label.
PRODUCTION
It is extracted from cocks' combs or obtained by fermentation from Streptococci, Lancefield Groups A and C. When produced by fermentation of gram-positive bacteria, the process must be shown to reduce or eliminate pyrogenic or inflammatory components of the cell wall.
CHARACTERS
Appearance: White or almost white, very hygroscopic powder or fibrous aggregate.
Solubility: Sparingly soluble or soluble in water, practically insoluble in acetone and in anhydrous ethanol.
IDENTIFICATION
A. Infrared absorption spectrophotometry. ComparisoniPh. Eur. reference spectrum of sodium hyaluronate.
B. It gives reaction of sodium.
TESTS
Solution S: Weigh a quantity of the substance to be examined equivalent to 0.10 g of the dried substance and add 30.0 ml of a 9 g/l solution of sodium chloride. Mix gently on a shaker until dissolved (about 12 h).
Appearance of solution: Solution S is clear and its absorbance at 600 nm is not greater than 0.01.
pH: 5.0 to 8.5.
Dissolve the substance to be examined in carbon dioxide-free water to obtain a solution containing a quantity equivalent to 5 mg of the dried substance per millilitre.
Intrinsic viscosity: To pass the test.
Sulphated glycosaminoglycans: Maximum 1 per cent, if the product is extracted from cocks' combs.
Nucleic acids: The absorbance of solution S at 260 nm is maximum 0.5.
Protein: Maximum 0.3 per cent; maximum 0.1 per cent, if intended for use in the manufacture of parenteral preparations.
Chlorides: Maximum 0.5 per cent.
Iron: Maximum 80.0 ppm.
Heavy metals: Maximum 20 ppm; maximum 10 ppm if intended for use in the manufacture of parenteral preparations.
Loss on drying: Maximum 20.0 per cent, determined on 0.500 g by drying at 100-110C over diphosphorus pentoxide for 6 h.
Microbial contamination: Total viable aerobic count not more than 100 micro-organisms per gram. Use 1 g of the substance to be examined.
Bacterial endotoxins: Less than 0.5 IU/mg, if intended for use in the manufacture of parenteral preparations without a further appropriate procedure for the removal of bacterial endotoxins; less than 0.05 IU/mg, if intended for use in the manufacture of intra-ocular preparations or intra-articular preparations without a further appropriate procedure for the removal of bacterial endotoxins.
Sodium Hyaluronate USP NF Grade and Purified Sodium Hyaluronate JP Grade can also be offered.
The Uses of Sodium Hyaluronate:
Sodium Hyaluronate is used to protect skin ulcers, burns, or wounds from irritation so that the skin can heal properly. Some products are also used to help relieve itching and pain from various skin conditions. It is also used to treat knee pain in patients with joint inflammation. When applied topically, sodium hyaluronate is excellent for reducing dryness and wrinkles. You can find it in products like serums, eye creams, etc.
The MSDS-SDS Hazard Statement of Sodium Hyaluronate:
Sodium Hyaluronate SDS, Material Safety Data Sheet
MSDS Sheet, Material Safety Data Sheet
1. PRODUCT DESCRIPTION
Product Name & Other Names: Sodium Hyaluronate.
CAS Number: 9067-32-7
EINECS EC Number: NA
Molecular Weight: 799.641
Molecular Formula: C28H44N2NaO23
Relevant uses and uses advised against (if any): Industrial Manufacturing.
2. HAZARD IDENTIFICATION
GHS, Globally Harmonized System Classification in accordance with 29 CFR 1910
Classification according to Regulation (EC) No 1272/2008
Not a hazardous substance or mixture according to Regulation (EC) No. 1272/2008.
This substance is not classified as dangerous according to Directive 67/548/EEC.
Labeling according to GHS & Regulation (EC) No 1272/2008
| GHS Label Elements None |
Signal Words: None
Precautionary statements:
P261: Avoid breathing dust/fume/gas/mist/vapors/spray.
P262: Do not get in eyes, on skin, or on clothing.
P281: Use personal protective equipment as required.
P302+P352: IF ON SKIN: Wash with plenty of soap and water.
P304+P340: IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing.
P337+313: If eye irritation persists get medical advice/attention.
3. COMPOSITION/INFORMATION ON INGREDIENTS
Product Name & Other Names: Sodium Hyaluronate.
CAS Number: 9067-32-7
4. FIRST AID MEASURES
Always get medical attention after the first aid is over.
Emergency and First Aid Procedures:
Eyes - Flush with water for at least 15 minutes, raising and lowering eyelids occasionally. Get medical attention if irritation persists.
Skin - Thoroughly wash exposed area for at least 15 minutes. Remove contaminated clothing. Launder contaminated clothing before reuse. Get medical attention if irritation persists.
Ingestion - If swallowed, if conscious, give plenty of water. Immediately call a physician. Never give anything by mouth to an unconscious person.
Inhalation - Remove to fresh air. Give oxygen if breathing is difficult; give artificial respiration if breathing has stopped. Get medical attention.
5. FIREFIGHTING PROCEDURES
Extinguisher Media: Use media suitable to extinguish surrounding fire. Use water spray, alcohol-resistant foam, dry chemical, or carbon dioxide.
Flammable Limits in Air % by Volume: N/A
Special Information: In the event of a fire, wear full protective clothing and NIOSH-approved self-contained breathing apparatus with full face piece operated in the pressure demand or other positive pressure mode. At high temperatures under fire conditions, it may produce toxic or irritating fumes. Fire-extinguishing work is done from the windward and the suitable fire-extinguishing method according to the surrounding situation is used.
6. SPILL OR LEAK PROCEDURES
Personal precautions, protective equipment, and emergency procedures: Avoid breathing dust/fumes/gas/mist/vapors/spray. Use individual protective equipment (waterproof boots, suitable protective clothing, safety glasses, etc.). Do not approach facing the wind.
Environmental precautions: Do not let the product enter drains, soil, or water sources.
Methods and materials used for containment Cleanup procedures and Storage: Do not inhale vapors, mist, or gas. Avoid dust formation. Contain spilled material. Cover with an inert, non-combustible absorbent material, (e.g. sand, earth, diatomaceous earth, vermiculite). Use a shovel to put the material into a convenient waste disposal container.
7. SPECIAL PRECAUTIONS
Precautions for safe handling: Apply according to good manufacturing and industrial hygiene practices. Ensure proper ventilation. In case of insufficient ventilation, wear suitable respiratory equipment. Wash thoroughly after handling. Do not drink, eat, or smoke while handling. Avoid contact with skin, eyes, and clothing. Minimize dust generation. Avoid breathing dust/fumes/gas/mist/vapors/spray. Keep container tightly closed. Avoid ingestion and inhalation. Use individual protective equipment (waterproof boots, suitable protective clothing, safety glasses, etc.).
Conditions for safe storage, including any incompatibilities: Store in cool, dry, and ventilated area away from heat sources and protected from sunlight in tightly closed original container. Keep air contact to a minimum. Store protected from heat, sparks and ignition sources and incompatible materials. Avoid contact with skin and eyes. Avoid inhalation of dust/mist/vapor. Do not store with incompatible materials like strong oxidizing agents.
8. SPECIAL PROTECTION INFORMATION
Exposure Guidelines: This product does not contain any hazardous materials with occupational exposure limits established by the region-specific regulatory bodies.
Ventilation System: A system of local and/or general exhaust is recommended to keep employee exposures as low as possible.
Personal Respirators (NIOSH Approved): For conditions of use where exposure to dust or mist is apparent and engineering controls are not feasible, a particulate respirator may be worn.
Skin Protection: Wear protective gloves and clean body-covering clothing.
Eye Protection: Use chemical safety goggles and/or full face shield where dusting or splashing of solutions is possible. Maintain eye wash fountain and quick-drench facilities in work area.
Other Control Measures: Maintain good housekeeping in work area. Handle in accordance with good industrial hygiene and safety practice.
9. PHYSICAL DATA
Appearance and Odor: It is white crystalline powder.
Odor: Characteristic odor.
Odor threshold: Not available.
pH: Not available.
Relative density: Not available.
Melting point/freezing point: Not available.
Initial boiling point and boiling range: Not available.
Flash point: Not available.
Auto-ignition temperature: Not available.
Decomposition temperature: Not available.
Upper/lower flammability or explosive limits: Not available.
Vapor pressure: Not available.
Vapor density: Not available.
Evaporation rate: Not available.
Flammability (solid, gas): Not available.
Partition coefficient: n-octanol/water: Not available.
Solubility in Water: Soluble.
Viscosity: Not available.
10. REACTIVITY DATA
Stability: It is stable
Conditions to Avoid: Heat, Direct sun light, Humidity, Dust generation.
Incompatibility (Materials to Avoid): Strong oxidizing agents.
Hazardous Decomposition Products: Carbon monoxide, carbon dioxide
Hazardous Polymerization: Will not occur.
11. TOXICITY DATA
Toxicity Data: Acute oral toxicity LD50 Rat: > 800 mg/kg
Carcinogenicity: No component of this product present at levels greater than or equal to 0.1% is identified as possible or confirmed human carcinogen by IARC, ACGIH, OSHA and NTP.
Teratogenic Effects: Not available.
Mutagenic Effects: Not available.
Developmental Toxicity: Not available.
Reproductive Effects: No information available.
12. ECOLOGICAL DATA
Toxicity to fish: No data available
Results of PBT and vPvB assessment: This substance/mixture contains no components considered to be either persistent, bioaccumulative and toxic (PBT), or very persistent and very bioaccumulative (vPvB) at levels of 0.1% or higher.
13. DISPOSAL INFORMATION
Waste Disposal Methods: Dispose in accordance with all applicable Federal, State and Local regulations.
14. TRANSPORT INFORMATION
DOT USA, TDG Canada & ADR/RID Europe : Not regulated.
IMDG/IMO: Not regulated.
IATA: Not regulated.
15. REGULATORY INFORMATION
SARA 311/312: See section 2.
California Prop 65 Components: Not listed.
Disclaimer:
**************************
Our company provides this MSDS sheet in good faith but makes no representation as to its comprehensiveness or accuracy. This SDS sheet is intended only as a guide to the appropriate precautionary handling of the material by a properly trained person using this product. The above information has been compiled from various sources and has the possibility of discrepancy and being out-dated information. Individuals receiving the information must exercise their independent judgment and do further search in determining its appropriateness for a particular purpose. In no case shall our company be liable to loss or damages by the product user.
**************************
Anmol Chemicals Industries Pvt. Ltd. is an off-shoot of Anmol Chemicals and Anmol Chemicals & Pharmaceuticals, Taloja. It is located in MIDC Taloja and it is manufacturing pharmaceutical grades of API, Excepients, Food grade and Reagent grade chemicals. Anmol Chemicals Industries Pvt. Ltd. is a several decades old group of companies, engaged in manufacturing, supplying, distributing, wholesale supplies for actual users, retail or small pack supplies for research and development chemicals, fine and speciality chemicals, pharmaceutical excipients, mineral fortifiers in chemically pure, Analytical reagent grade, IP BP USP Ph Eur EP JP and other pharmaceutical grade monograph including FCC Food grade chemicals and Nutraceuticals, Mineral Fortifiers at best prices.
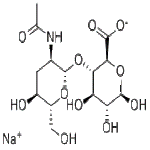
Sodium Hyaluronate Structure
CAS Number 9067-32-7, Sodium Hyaluronate Manufacturer Exporter
ANMOL CHEMICALS INDUSTRIES Pvt. Ltd.
India, USA, Europe, UAE
TELEPHONE: 91-22-65290110
Navi Mumbai, INDIA
e-mail: info(At the rate i.e. @)anmol.org
Copyright. 4-apr-26
We manufacture:
Glacial Acetic Acid Manufacturer
Hydrated Manganese Glycerophosphate
Nebivolol Hydrochloride or Nebivolol HCl

