CAS Number 557-04-0, Magnesium Stearate USP NF BP Ph Eur FCC Food Grade Manufacturers Exporters







CAS Number 557-04-0, Magnesium Stearate Manufacturer Exporter
For Properties Specifications of Magnesium Stearate Click Properties, Specifications of Magnesium Stearate Manufacturer.
For Uses of Magnesium Stearate Click Uses of Magnesium Stearate Manufacturer.
For For SDS MSDS Sheet of Magnesium Stearate Click SDS Safety Data Sheet MSDS Sheet of Magnesium Stearate Manufacturer.
The Properties and Specifications of Magnesium Stearate:
Magnesium Stearate USP NF Grade Specifications
Octadecanoic acid, magnesium salt. ----- CAS 557-04-0
Magnesium Stearate is a compound of magnesium with a mixture of solid organic acids, and consists chiefly of variable proportions of magnesium stearate and magnesium palmitate. The fatty acids are derived from edible sources. It contains not less than 4.0 percent and not more than 5.0 percent of Mg, calculated on the dried basis.
Identification:
A: Mix 5.0 g Magnesium stearate with 50 mL of peroxide-free ether, 20 mL of diluted nitric acid, and 20 mL of water in a round-bottom flask. Connect the flask to a reflux condenser, and reflux until dissolution is complete. Allow to cool, and transfer the contents of the flask to a separator. Shake, allow the layers to separate, and transfer the aqueous layer to a flask. Extract the ether layer with two 4-mL portions of water, and add these aqueous extracts to the main aqueous extract. Wash the aqueous extract with 15 mL of peroxide-free ether, transfer the aqueous extract to a 50-mL volumetric flask, dilute with water to volume, and mix. Retain this solution for the Limit of chloride and Limit of sulfate tests. This solution responds to the test for Magnesium.
B: The retention times of the peaks corresponding to stearic acid and palmitic acid in the chromatogram of the Test solution correspond to those in the chromatogram of the System suitability solution, as obtained in the Relative content of stearic acid and palmitic acid test.
Microbial limits: The total aerobic microbial count does not exceed 1000 cfu per g, the total combined molds and yeasts count does not exceed 500 cfu per g, and it meets the requirements of the tests for absence of Salmonella species and Escherichia coli.
Acidity or alkalinity: Transfer 1.0 g Magnesium stearate to a 100-mL beaker, add 20 mL of carbon dioxide-free water, boil on a steam bath for 1 minute with continuous shaking, cool, and filter. Add 0.05 mL of bromothymol blue TS to 10 mL of the filtrate: not more than 0.05 mL of 0.1 N hydrochloric acid or 0.1 N sodium hydroxide is required to change the color of the indicator.
Loss on drying: Dry it at 105 to constant weight: it loses not more than 6.0% of its weight.
Specific surface area: [NOTE—In cases where there are no functionality-related concerns regarding the specific surface area of this article, this test may be omitted.] Where the labeling states the specific surface area, determine the specific surface area value as directed in the chapter in the P/P0 range of 0.05 to 0.15, and using out gassing conditions of 2 hours at 40 . If the plot deviates from linearity for P/P0 values of 0.05 to 0.15, then a suitable range of P/P0 values should be validated for linearity. In this case, it is necessary to state the range of validated P/P0 values, the increment of the P/P0 values, and the out gassing conditions employed.
Limit of chloride: A 10.0-mL portion of the aqueous solution obtained in Identification test A shows no more chloride than corresponds to 1.4 mL of 0.020 N hydrochloric acid (0.1%).
Limit of sulfate: A 3.0 mL portion of the aqueous solution obtained in Identification test A shows no more sulfate than corresponds to 3.0 mL of 0.020 N sulfuric acid (1.0%).
Lead: Ignite 0.50 g Magnesium stearate in a silica crucible in a muffle furnace at 475 to 500 for 15 to 20 minutes. Cool, add 3 drops of nitric acid, evaporate over a low flame to dryness, and again ignite at 475 to 500 for 30 minutes. Dissolve the residue in 1 mL of a mixture of equal parts by volume of nitric acid and water, and wash into a separator with several successive portions of water. Add 3 mL of Ammonium Citrate Solution and 0.5 mL of Hydroxylamine Hydrochloride Solution, and render alkaline to phenol red TS with ammonium hydroxide. Add 10 mL of Potassium Cyanide Solution. Immediately extract the solution with successive 5-mL portions of Dithizone Extraction Solution, draining off each extract into another separator, until the last portion of dithizone solution retains its green color. Shake the combined extracts for 30 seconds with 20 mL of 0.2 N nitric acid, and discard the chloroform layer. Add to the acid solution 4.0 mL of Ammonia-Cyanide Solution and 2 drops of Hydroxylamine Hydrochloride Solution. Add 10.0 mL of Standard Dithizone Solution, and shake the mixture for 30 seconds. Pass the chloroform layer through an acid-washed filter paper into a color-comparison tube, and compare the color with that of a standard solution prepared as follows. To 20 mL of 0.2 N nitric acid add 5 ?g of lead, 4 mL of Ammonia-Cyanide Solution, and 2 drops of Hydroxylamine Hydrochloride Solution, and shake with 10.0 mL of Standard Dithizone Solution for 30 seconds. Pass through an acid-washed filter paper into a color comparison tube. The color of the sample solution does not exceed that of the control (0.001%).
Relative content of stearic acid and palmitic acid: The stearate peak comprises not less than 40%; and the sum of the stearate and palmitate peaks is not less than 90% of the total area of all fatty acid ester peaks in the chromatogram.
Magnesium Stearate BP Ph Eur Grade Specifications
DEFINITION
Magnesium stearate is a mixture of magnesium salts of different fatty acids consisting mainly of stearic (octadecanoic) acid [(C17H35COO)2Mg] and palmitic (hexadecanoic) acid [(C15H31COO)2 Mg] with minor proportions of other fatty acids. It contains not less than 4.0 per cent and not more than 5.0 per cent of Mg, calculated with reference to the dried substance. The fatty acid fraction contains not less than 40.0 per cent of stearic acid and the sum of stearic acid and palmitic acid is not less than 90.0 per cent.
CHARACTERS
A white or almost white, very fine, light powder, greasy to the touch, practically insoluble in water and in ethanol.
IDENTIFICATION
First identification C, D.
Second identification A, B, D.
A. The residue obtained in the preparation of solution S (see Tests) has a freezing point not lower than 53C.
B. The acid value of the fatty acids (2.5.1) is 195 to 210, determined on 0.200 g of the residue obtained in the preparation of solution S dissolved in 25 ml of the prescribed mixture of solvents.
C. Examine the chromatograms obtained in the test for fatty acid composition. The retention times of the principal peaks in the chromatogram obtained with the test solution are approximately the same as those of the principal peaks in the chromatogram obtained with the reference solution.
D. 1 ml of solution S gives the reaction of magnesium.
TESTS
Solution S: To 5.0 g Magnesium stearate add 50 ml of peroxide-free ether, 20 ml of dilute nitric acid and 20 ml of distilled water and heat under a reflux condenser until dissolution is complete. Allow to cool. In a separating funnel, separate the aqueous layer and shake the ether layer with 2 quantities, each of 4 ml, of distilled water. Combine the aqueous layers, wash with 15 ml of peroxide-free ether and dilute to 50 ml with distilled water R (solution S). Evaporate the organic layer to dryness and dry the residue at 100-105C. Keep the residue for identification tests A and B.
Acidity or alkalinity: To 1.0 g Magnesium stearate add 20 ml of carbon dioxide-free water R and boil for 1 min with continuous stirring. Cool and filter. To 10 ml of the filtrate add 0.05 ml of bromothymol blue solution. Not more than 0.5 ml of 0.01 M hydrochloric acid or 0.01 M sodium hydroxide is required to change the colour of the indicator.
Chlorides: 0.5 ml of solution S diluted to 15 ml with water R complies with the limit test for chlorides (0.1 per cent).
Sulphates: 0.3 ml of solution S diluted to 15 ml with distilled water complies with the limit test for sulphates (0.5 per cent).
Cadmium: Not more than 3.0 ppm of Cd, determined by atomic absorption spectrometry.
Lead: Not more than 10.0 ppm of Pb, determined by atomic absorption spectrometry.
Nickel: Not more than 5.0 ppm of Ni, determined by atomic absorption.
Loss on drying: Not more than 6.0 per cent, determined on 1.000 g by drying in an oven at 105C.
Microbial contamination: Total viable aerobic count not more than 103 micro-organisms per gram, determined by plate count. It complies with the test for Escherichia coli.
Magnesium Stearate FCC Food Grade Specifications
Magnesium stearate CAS 557-04-0
DESCRIPTION
Magnesium Stearate occurs as a fine, white, bulky powder that is unctuous and free from grittiness. It is a compound of magnesium with a mixture of solid organic acids obtained from edible sources and consists chiefly of variable proportions of Magnesium Stearate and magnesium palmitate. It is insoluble in water, in alcohol, and in ether.
Function: Ant caking agent; binder; emulsifier.
REQUIREMENTS
Identification:
A. Heat 1 g of Magnesium stearate with a mixture of 25 mL of water and 5 mL of hydrochloric acid. Liberated fatty acids float as an oily layer on the surface of the liquid. The water layer gives positive tests for Magnesium.
B. Mix 25 g of Magnesium stearate with 200 mL of hot water, then add 60 mL of 2 N sulfuric acid, and while stirring frequently, heat the mixture until the fatty acids separate cleanly as a transparent layer. Wash the fatty acids with boiling water until they are free from sulfate, collect them in a small beaker, and warm them on a steam bath until the water has separated and the fatty acids are clear. Allow the acids to cool, pour off the water layer, then melt the acids, filter into a dry beaker, and dry at 105C for 20 min. The solidification point of the fatty acids so obtained is not below 54C
Assay: Not less than 6.8% and not more than 8.3% of MgO.
Lead: Not more than 5 mg/kg.
Loss on Drying: Not more than 4.0%.
The Uses of Magnesium Stearate:
Magnesium Stearate is widely used for many decades in the food industry as an emulsifier, binder and thickener, as well as an anticaking, lubricant, release, and antifoaming agent. It is present in many food supplements, confectionery, chewing gum, herbs and spices, and baking ingredients. Magnesium stearate is an additive that's primarily used in medication capsules. It's considered a “flow agent”. Magnesium stearate is widely used in cosmetics, foods, and pharmaceutical formulations.
The MSDS-SDS Hazard Statement of Magnesium Stearate:
Magnesium Stearate SDS, Safety Data Sheet
MSDS Sheet, Material Safety Data Sheet
1. PRODUCT NAME AND COMPANY IDENTIFICATION
Product Name & Other Names: Magnesium Stearate.
CAS #: 557-04-0
EINECS EC Number: 209-150-3
Chemical Formula: (C17H35COO)2Mg
Molecular Weight: 591.24
Relevant uses and uses advised against (if any): Laboratory and Industrial Manufacturing.
2. HAZARDS IDENTIFICATION
GHS, Globally Harmonized System Classification in accordance with 29 CFR 1910
Classification according to Regulation (EC) No 1272/2008
Not a hazardous substance or mixture according to Regulation (EC) No. 1272/2008.
This substance is not classified as dangerous according to Directive 67/548/EEC.
Labeling according to GHS & Regulation (EC) No 1272/2008
| GHS Label Elements NONE |
Signal Word: None
Precautionary statements:
P261: Avoid breathing dust/fume/gas/mist/vapors/spray.
P262: Do not get in eyes, on skin, or on clothing.
P281: Use personal protective equipment as required.
P302+P352: IF ON SKIN: Wash with plenty of soap and water.
P304+P340: IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing.
P337+313: If eye irritation persists get medical advice/attention.
3. COMPOSITION/INGREDIENT INFORMATION
Product Name & Other Names: Magnesium Stearate.
CAS #: 557-04-0
EINECS EC Number: 209-150-3
4. FIRST AID MEASURES
Eyes: Flush with plenty of water or eye wash solution for 15 minutes. WARM water MUST be used. Get medical attention if irritation persists.
Skin: Wash with soap and water. Cover irritated skin with an emollient. Get medical attention if irritation occurs.
Ingestion: Do NOT induce vomiting unless directed to do so by medical personnel. Never give anything by mouth to an unconscious person. If large quantities of this material are swallowed, call a physician immediately. Loosen tight clothing such as collars, ties, belts, or waistbands.
Inhalation: Remove to fresh air. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Seek medical attention.
5. FIRE FIGHTING MEASURES
Flammability of Product: Magnesium stearate may be combustible at high temperatures.
Hazardous Combustion Products: CO, CO2 and MgO
Extinguishing Media: Dry Chemical; Carbon Dioxide; Foam Note: Do not use solid water jet.
Special Information: In the event of a fire, wear full protective clothing and NIOSH-approved self-contained breathing apparatus with full face piece operated in the pressure demand or other positive pressure mode. At high temperatures under fire conditions, it may produce toxic or irritating fumes. Fire-extinguishing work is done from the windward and the suitable fire-extinguishing method according to the surrounding situation is used.
6. ACCIDENTAL RELEASE MEASURES
Personal precautions, protective equipment, and emergency procedures: Ventilate area of leak or spill. Avoid breathing dust/fumes/gas/mist/vapors/spray. Use individual protective equipment (waterproof boots, suitable protective clothing, safety glasses, etc.). Prevent any contact with hot surfaces. Do not approach facing the wind.
Environmental precautions: Do not let the product enter drains, soil, or water sources.
Methods and materials used for containment Cleanup procedures and Storage: Contain spilled material. Cover with an inert, non-combustible absorbent material, (e.g. sand, earth, diatomaceous earth, vermiculite). Vacuum or sweep-up and remove to an approved disposal container.
7. HANDLING AND STORAGE
Precautions for safe handling: Apply according to good manufacturing and industrial hygiene practices. Ensure proper ventilation. In case of insufficient ventilation, wear suitable respiratory equipment. Wash thoroughly after handling. Do not drink, eat, or smoke while handling. Avoid contact with skin, eyes, and clothing. Minimize dust generation. Avoid breathing dust/fumes/gas/mist/vapors/spray. Keep container tightly closed. Avoid ingestion and inhalation. Use individual protective equipment (waterproof boots, suitable protective clothing, safety glasses, etc.).
Conditions for safe storage, including any incompatibilities: Store in cool, dry, and ventilated area away from heat sources and protected from sunlight in tightly closed original container. Keep air contact to a minimum. Store protected from heat, sparks and ignition sources and incompatible materials. Avoid contact with skin and eyes. Avoid inhalation of dust/mist/vapor. Do not store with incompatible materials like strong oxidizing agents.
8. EXPOSURE CONTROL/PERSONAL PROTECTION
Engineering Controls: Use process enclosures. Provide exhaust ventilation or other engineering controls to keep the airborne concentrations of vapors below their respective threshold limit value.
Ventilation System : A system of local and/or general exhaust is recommended to keep employee exposures as low as possible.
Personal Respirators (NIOSH Approved): For conditions of use where exposure to dust or mist is apparent and engineering controls are not feasible, a particulate respirator may be worn.
Skin Protection: Wear protective gloves and clean body-covering clothing.
Eye Protection: Use chemical safety goggles and/or full face shield where dusting or splashing of solutions is possible. Maintain eye wash fountain and quick-drench facilities in work area.
Other Control Measures: Maintain good housekeeping in work area. Handle in accordance with good industrial hygiene and safety practice.
9. PHYSICAL AND CHEMICAL PROPERTIES
Appearance: White solid
Odor: Not available.
Odor threshold: Not available.
pH: Not available.
Relative density: around 1.03
Melting Point: 88C (190.4F)
Boiling Point: Not available.
Flash point: Not available.
Auto-ignition temperature: Not available.
Decomposition temperature: Not available.
Upper/lower flammability or explosive limits: Not available.
Vapor pressure: Not available.
Vapor density: Not available.
Evaporation rate: Not available.
Flammability (solid, gas): Not available.
Partition coefficient: n-octanol/water: Not available.
Solubility in Water: Slightly soluble in cold water.
Viscosity: Not available.
10. STABILITY AND REACTIVITY
Stability: Stable under normal usage.
Conditions to Avoid: Not Available
Incompatibility (Materials to Avoid): Strong oxidizing agents.
Hazardous Decomposition products: Magnesium oxide, carbon oxides and fumes.
Hazardous Polymerization: Will not occur.
11. TOXICOLOGICAL INFORMATION
Toxicity to Animals: LD50: Not Available
Carcinogenicity: No component of this product present at levels greater than or equal to 0.1% is identified as probable, possible or confirmed human carcinogen by IARC.
Reproductive Toxicity: Not Available
Mutagenic: Not Available.
12. ECOLOGICAL INFORMATION
Ecological Information: Not Available
Products of Biodegradation: Possibly hazardous short-term degradation products are not likely. However, long term degradation products may arise.
Results of PBT and vPvB assessment: This substance/mixture contains no components considered to be either persistent, bioaccumulative and toxic (PBT), or very persistent and very bioaccumulative (vPvB) at levels of 0.1% or higher.
13. DISPOSAL CONSIDERATIONS
Waste Disposal Methods: Do not put into sewer lines. Dispose of according to local, state regulations.
14. TRANSPORT INFORMATION
DOT USA, TDG Canada & ADR/RID Europe: Not controlled.
IMO/IMDG: Not controlled.
IATA/ICAO: Not controlled.
15. REGULATORY INFORMATION
USA Regulations:
Section 311/312: See section 2.
Section 313: None
DISCLAIMER: The information and recommendations set forth herein are presented in good faith and believed correct as of the date hereof. It is compiled from various sources and it is not necessarily all inclusive nor fully adequate in every circumstance. In addition, these suggestions should not be confused with nor followed in violation of applicable laws, regulations, rules or insurance requirements applicable. This MSDS sheet is intended only as a guide to the appropriate precautionary handling of the material by a properly trained person using this product. Individuals receiving the information must exercise their independent judgment in determining its appropriateness for a particular purpose.
Anmol Chemicals Industries Ltd. is an off-shoot of Anmol Chemicals and Anmol Chemicals & Pharmaceuticals, Taloja. It is located in MIDC Taloja and it is manufacturing pharmaceutical grades of API, Excepients, Food grade and Reagent grade chemicals. Anmol Chemicals Industries Pvt. Ltd. is a several decades old group of companies, engaged in manufacturing, supplying, distributing, wholesale supplies for actual users, retail or small pack supplies for research and development chemicals, fine and speciality chemicals, pharmaceutical excipients, mineral fortifiers in chemically pure, Analytical reagent grade, IP BP USP Ph Eur EP JP and other pharmaceutical grade monograph including FCC Food grade chemicals and Nutraceuticals, Mineral Fortifiers at best prices.
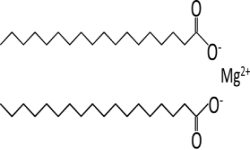
Magnesium Stearate Structure
CAS Number 557-04-0, Magnesium Stearate Manufacturer Exporter
ANMOL CHEMICALS INDUSTRIES Pvt. Ltd.
India, USA, Europe, UAE
TELEPHONE: 91-22-65290110
Navi Mumbai, INDIA
e-mail: info(At the rate i.e. @)anmol.org
Copyright. 25-may-26
We manufacture:
Glacial Acetic Acid Manufacturer
Biotin or Vitamin B7 or Vitamin H
Calcium Gluceptate or Calcium Glucoheptonate
Maltodextrin

